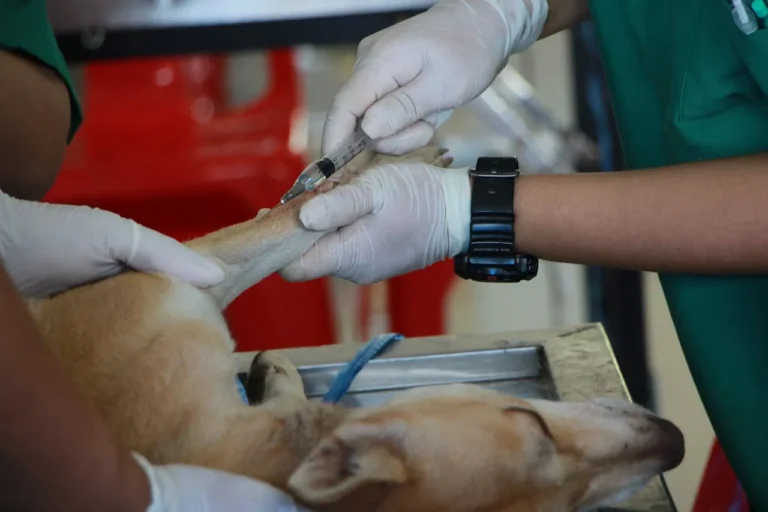
90% of Healthcare Providers Worldwide Express Concern Over Social Media’s Role in Spreading Fad Diets
The 40th barometer from Sermo, a fast, frictionless HCP engagement platform providing the healthcare industry with real-time business insights and authentic physician touch points, of 1,000+ global healthcare providers (HCPs) found that the majority (90%) are concerned about the promotion of…