Qure.ai’s Latest FDA Clearance Enhances Early Detection and Clinical Support in Chest Radiography
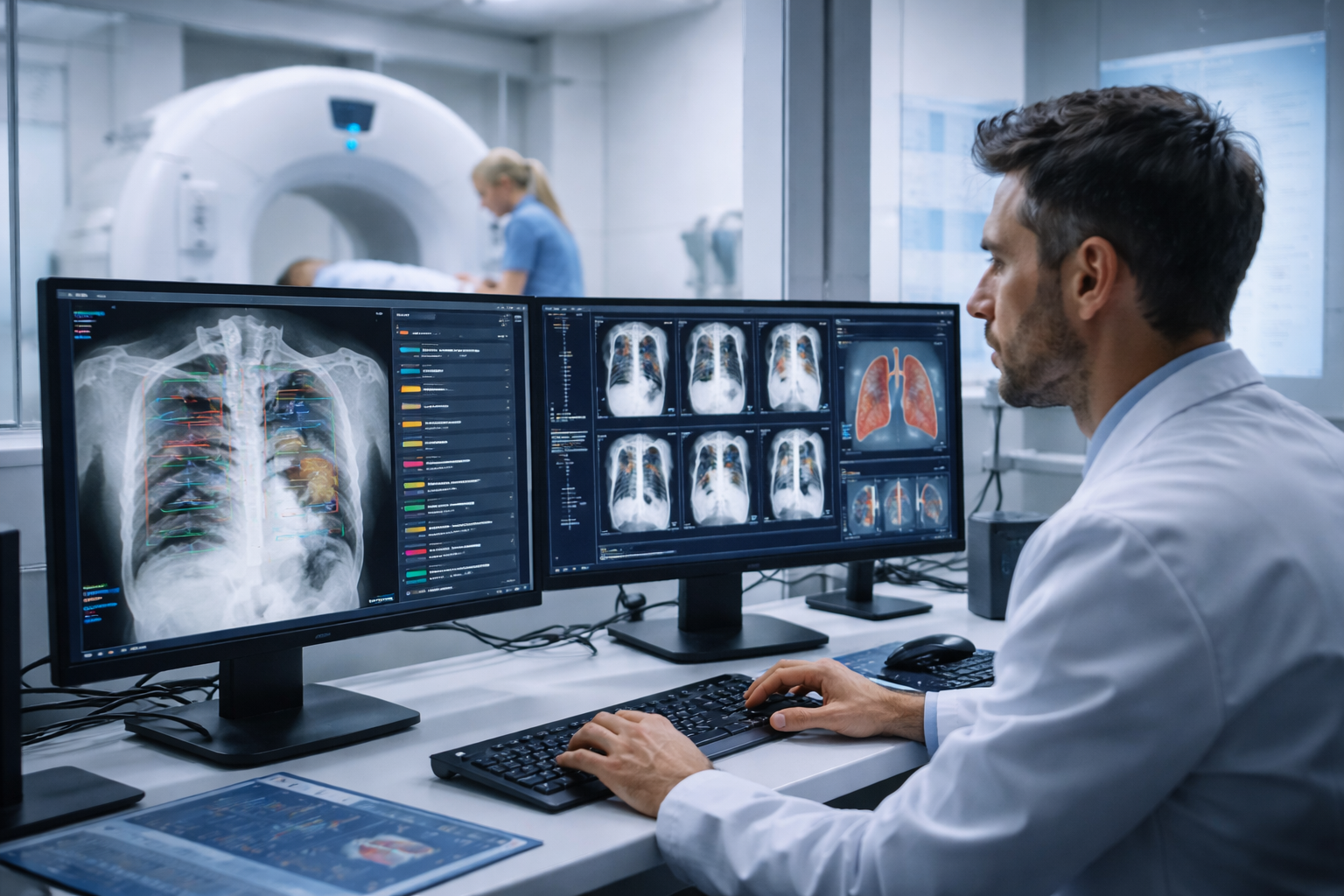
Why is the early detection of abnormalities in chest X-rays critical for patient outcomes? Qure.ai, a global leader in AI-driven healthcare solutions, has received 510(k) class II clearance from the US Food and Drug Administration (FDA) for its qXR-Detect, a computer-assisted detection (CADe) radiography solution. This clearance marks a significant advancement in the company’s mission to support clinicians and improve patient care through advanced AI technologies.
The FDA’s clearance of qXR-Detect is a pivotal moment for Qure.ai, as it now covers 26 total indications across nine products for X-ray and CT, with over 65 CE certified indications and other global validations. This latest clearance adds breadth to the company’s product portfolio in the US, enhancing its ability to identify and categorize key positive findings on plain film chest X-rays. According to Professor Amit Gupta, Division Chief of Cardiothoracic Imaging and Modality Director of Diagnostic Radiography at University Hospitals Cleveland Medical Center, “There are more than 100 potential findings on a plain film chest X-ray that AI tools can help detect and assist radiologists with, supporting earlier recognition of abnormalities.”
Key Insights at a Glance
- Early Detection: qXR-Detect enhances the early identification of abnormalities in chest X-rays.
- Comprehensive Coverage: The clearance covers six regions of interest, including lung, pleura, and bone.
- Regulatory Milestone: Qure.ai now has 26 FDA cleared indications, solidifying its position as a leader in AI healthcare.
- Clinical Support: The tool provides visual localization and explainability, aiding radiologists in their interpretations.
The Critical Need for Enhanced Chest X-Ray Analysis
Chest radiography is the most performed imaging exam in the US, with 70 million obtained annually in emergency rooms, outpatient departments, and routine health assessments. The complexity of chest X-rays, with over 100 potential findings, underscores the critical need for advanced AI tools to support clinicians. Early detection and accurate categorization of abnormalities can significantly improve patient outcomes, making this technology a vital asset in healthcare settings.
The Regulatory Clock Is Ticking for AI Innovations
Just as a skilled conductor guides an orchestra to produce a harmonious symphony, Qure.ai’s qXR-Detect orchestrates the detection and categorization of chest X-ray findings with precision and speed. The FDA clearance is a testament to the company’s rigorous adherence to regulatory standards and its commitment to delivering reliable, cutting-edge solutions. This clearance ensures that healthcare providers have access to the most up-to-date and accurate AI algorithms, enhancing their ability to diagnose and treat patients effectively.
Qure.ai’s qXR-Detect: A Game-Changer in Chest Radiography
Qure.ai has achieved a significant milestone with the FDA clearance of qXR-Detect, the only chest X-ray CADe device cleared by the FDA with a Predetermined Change Control Plan (PCCP). This plan ensures that US health system customers have access to the most current version of the algorithm as it evolves. According to Samir Shah, Chief Medical Officer at Qure.ai, “These super six indications cleared by the FDA for qXR-Detect establishes Qure.ai as the foremost commercial AI company, with the most comprehensive regulatory clearances for chest X-ray globally.”
The device’s performance was validated through a standalone and multi-reader multi-case (MRMC) clinical study, meeting predefined success criteria. This robust validation process underscores the reliability and effectiveness of qXR-Detect in supporting clinicians in their daily practice.
Future Outlook
The future of AI in healthcare is rapidly evolving, and Qure.ai’s latest FDA clearance is a clear indication of the company’s commitment to innovation and regulatory compliance. As the company continues to expand its product portfolio and global reach, the integration of advanced AI technologies in clinical settings will become increasingly prevalent. The next milestone for Qure.ai is to further enhance its AI algorithms and expand its regulatory clearances to cover a broader range of medical imaging applications.
Conclusion
The FDA clearance of qXR-Detect by Qure.ai represents a significant step forward in the early detection and management of chest X-ray abnormalities. For healthcare providers, this technology offers a powerful tool to improve patient outcomes and streamline clinical workflows. How is your healthcare organization preparing to integrate AI-driven solutions like qXR-Detect into your practice?
About Qure.ai
Qure.ai is a global AI company innovating solutions in healthcare for early detection and care management. Qure’s solutions power early detection of lung nodules, neurocritical findings and infectious diseases to support clinicians and propel developments in the pharmaceutical and medical device industries. The company empowers healthcare workers and health systems by helping to identify pathology quickly, prioritize treatment planning, and ultimately improve quality of patient lives.
Source link: https://www.businesswire.com/