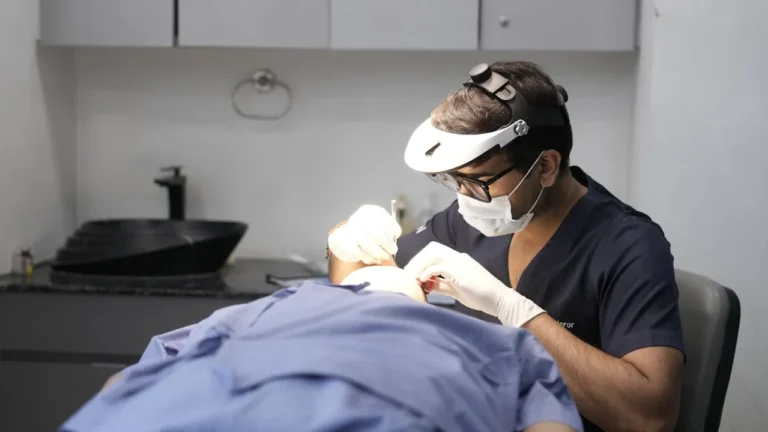
Lundbeck Shares AAN 2026 Data on Cognitive Symptom Improvement with VYEPTI in Migraine Patients
Lundbeck Shares AAN 2026 Data Showing Real-World Cognitive Improvements in Migraine Patients Treated with VYEPTI Lundbeck today presented new real-world 6-month results from the INFUSE study at the American Academy of Neurology (AAN) 2026 Annual Meeting, underscoring the importance of evaluating the…