
Simple Blood Test Can Predict Future Heart Attacks and Cardiac Events
Siemens Healthineers has announced a new prognostic claim for its Atellica IM High-Sensitivity Troponin I (TnIH) test, significantly enhancing care for at-risk cardiac patients in the U.S. This straightforward blood test allows healthcare providers to identify patients at risk of…

Airway Therapeutics Launches Phase 3 Study of Zelpultide Alfa for Preterm Infants at Risk of BPD
Airway Therapeutics, Inc. has announced the launch of a multinational Phase 3 clinical trial for zelpultide alfa (rhSP-D). This study aims to prevent bronchopulmonary dysplasia (BPD) and mitigate lung injury in premature infants. The biopharmaceutical company focuses on developing innovative…

Invisalign® Palatal Expander Now Available in Hong Kong for Dental Growth
Align Technology, Inc. (Nasdaq: ALGN), a global leader in medical devices known for its Invisalign® clear aligners, iTero™ intraoral scanners, and exocad™ CAD/CAM software, has announced the launch of its Invisalign Palatal Expander System in Hong Kong. This system is…

MEDiC Life Sciences Secures $5M Investment from LG and Hanmi for Cancer Biomarker Technology
MEDiC Life Sciences, a prominent functional genomics startup in Silicon Valley, has announced a $5M strategic investment from LG and Hanmi Pharmaceutical. This funding precedes MEDiC’s upcoming Series-A round later this year. Previously, the company raised $8M in a Seed…
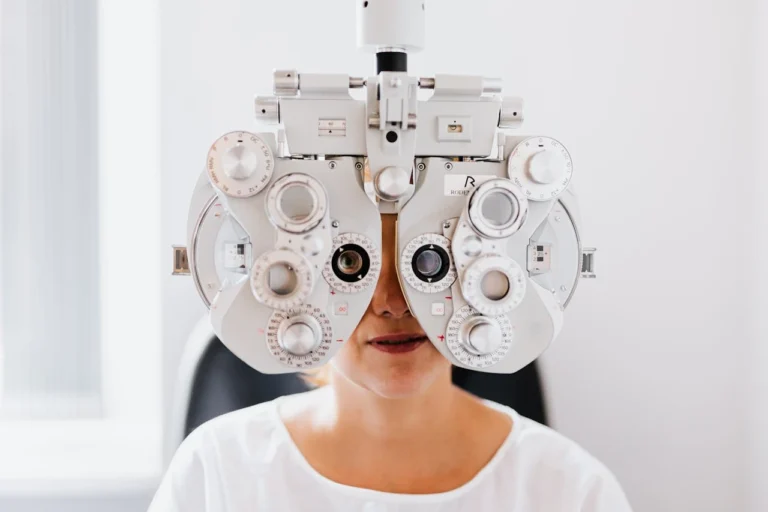
FDA Clears SPECTRALIS Flex Module for Enhanced Ophthalmic Imaging Access
Heidelberg Engineering, a leader in ophthalmic imaging and healthcare data solutions, has announced FDA clearance for the SPECTRALIS® Flex Module. This innovative multimodal platform is specifically designed for diagnostic imaging of the posterior segment in both pediatric and adult patients…

MEDiC Life Sciences Partners with Hanmi Pharmaceutical on Cancer Biomarker Discovery
MEDiC Life Sciences, a Silicon Valley biotech startup, has announced a research collaboration with Hanmi Pharmaceutical, a prominent biopharma company based in Korea. Through this partnership, MEDiC will utilize its MCAT™ platform to identify cancer biomarkers related to one of…

Exelixis and Merck Collaborate on Zanzalintinib Trials for Cancer
Exelixis, Inc. (Nasdaq: EXEL) and Merck (NYSE: MRK), known as MSD outside the U.S. and Canada, have announced a clinical development collaboration. The partnership will evaluate the investigational tyrosine kinase inhibitor (TKI) zanzalintinib in combination with Merck’s anti-PD-1 therapy KEYTRUDA®…

Bausch + Lomb Gets FDA Approval for enVista® Envy™ Intraocular Lens
Bausch + Lomb Corporation (NYSE/TSX: BLCO), a leading global eye health company, has announced that the U.S. Food and Drug Administration (FDA) has approved the enVista® Envy™ intraocular lens (IOL), which provides a continuous range of vision with excellent tolerance…

GE HealthCare Launches User-Friendly Versana Premier Ultrasound System
GE HealthCare (Nasdaq: GEHC) has introduced the Versana Premier™, the latest addition to its Versana ultrasound family. This system is designed to be reliable, affordable, and user-friendly, offering versatile features for healthcare professionals. Equipped with automation and AI-powered productivity tools,…

Most SMA Children on Evrysdi Can Sit, Stand, and Walk Independently: Two-Year Study
Genentech, part of the Roche Group, shared encouraging two-year data from the ongoing RAINBOWFISH study at the 29th World Muscle Society Congress (October 8-12, 2024). This study evaluated the efficacy and safety of Evrysdi® (risdiplam) in children with spinal muscular…

FDA Approves FoundationOne® Liquid CDx for Itovebi™ in Breast Cancer with PIK3CA Mutation
Foundation Medicine, Inc. has announced FDA approval for FoundationOne® Liquid CDx as a companion diagnostic for Itovebi™ (inavolisib) when used with palbociclib (Ibrance®) and fulvestrant. This therapy, developed by Genentech, is designed for adult patients with endocrine-resistant, PIK3CA-mutated, hormone receptor-positive,…

FDA Approves Pfizer’s HYMPAVZI™ for Hemophilia A and B Treatment
Pfizer Inc. (NYSE: PFE) has announced that the U.S. Food and Drug Administration (FDA) has approved HYMPAVZI™ (marstacimab-hncq) for routine prophylaxis. This treatment is designed to prevent or reduce the frequency of bleeding episodes in adults and pediatric patients aged…

