
HCA Healthcare Appoints Wendy Warren as SVP & Chief Ethics Officer
HCA Healthcare, Inc. (NYSE: HCA), a leading healthcare provider in the United States, has announced the appointment of Wendy Warren as senior vice president and chief ethics and compliance officer, effective March 3, 2025. Warren, who has been with the…
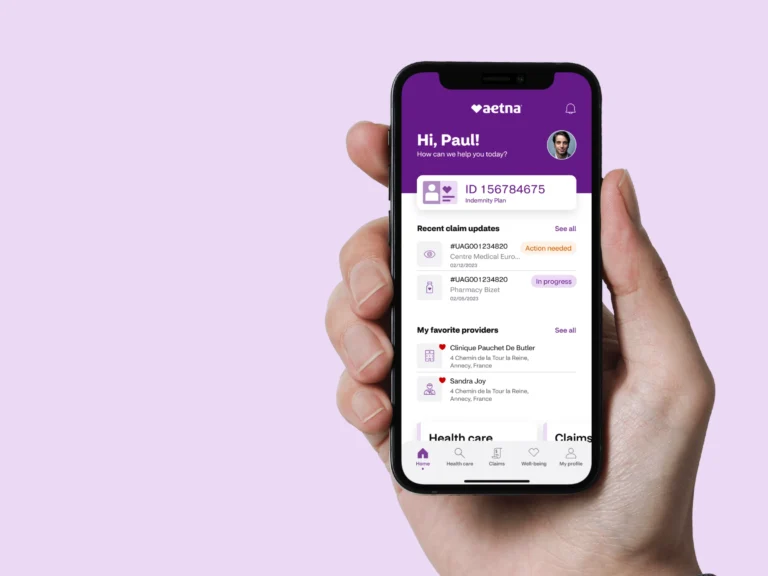
Aetna Enhances Health App with Spanish Language Support
Aetna®, a CVS Health® company (NYSE: CVS), has enhanced the Aetna Health℠ app by adding Spanish language capabilities, aiming to improve access to care for Spanish-speaking health plan members nationwide. This update allows eligible members, regardless of their coverage type,…

Exicure Announces Approval of $8.7 Million Equity Financing and Executive/Board Changes
Exicure, Inc. (Nasdaq: XCUR, “Exicure” or the “Company”), today announced that its stockholders approved a second investment of $8.7 million from HiTron Systems Inc. (“HiTron”), a publicly listed company on the KOSPI exchange in South Korea. This approval was in…

Aptorum Group Reports 2024 Mid-Year Financial Results & Business Update
Aptorum Group Limited (NASDAQ: APM), a clinical-stage biopharmaceutical company focused on addressing unmet medical needs in oncology, autoimmune, and infectious diseases, today released its business update and financial results for the six months ending June 30, 2024. Business Update Aptorum…

Vertex Announces FDA Approval of ALYFTREK™ for Cystic Fibrosis Treatment
Vertex Pharmaceuticals Incorporated (Nasdaq: VRTX) has announced that the U.S. Food and Drug Administration (FDA) has approved ALYFTREK (vanzacaftor/tezacaftor/deutivacaftor), a once-daily, next-generation CFTR (cystic fibrosis transmembrane conductance regulator) modulator. ALYFTREK is approved for the treatment of cystic fibrosis (CF) in…

FDA Approves Pfizer’s BRAFTOVI® for First-Line BRAF V600E-Mutant Colorectal Cancer
Pfizer Inc. (NYSE: PFE) has announced that the U.S. Food and Drug Administration (FDA) has granted approval for BRAFTOVI® (encorafenib) in combination with cetuximab (marketed as ERBITUX®) and mFOLFOX6 (fluorouracil, leucovorin, and oxaliplatin) as a treatment for patients with metastatic…

JCR Pharmaceuticals Launches Phase III Trial of JR-142, Long-Acting Growth Hormone Therapy
JCR Pharmaceuticals Co., Ltd. (TSE 4552) today announced the dosing of the first patient in its Phase III clinical trial of JR-142 (INN: redalsomatropin alfa) in Japan. This marks a significant milestone for the company in the development of JR-142,…

ZIVO Bioscience Reports Early Efficacy of Proprietary Ingredients Against Avian Influenza
ZIVO Bioscience, Inc. (OTCQB: ZIVO), a leading biotech and agtech R&D company focused on developing therapeutic, medicinal, and nutritional products derived from proprietary algal cultures, has announced positive preliminary results from a recent study. Conducted in collaboration with the University…

Quantum-Si Unveils Advances in Protein Variant Detection with ProteoVue™ Software
Quantum-Si Incorporated (Nasdaq: QSI), known as The Protein Sequencing Company™, has announced the submission of a new preprint paper to BioRxiv, showcasing the power of its proprietary bioinformatics tool, ProteoVue, in detecting and quantifying Single Amino Acid Variants (SAAVs). The…

Bruker Unveils 1.2 GHz NMR Spectrometer at Swiss Facility
Bruker has announced the installation of a 1.2 GHz Ascend™ Nuclear Magnetic Resonance (NMR) spectrometer at the Swiss High-field NMR Facility, a collaborative research center jointly operated by the University of Basel, ETH Zürich, and the University of Zürich. This…

MIRAILAB BIOSCIENCE Launches NMN Supplements and Cosmetics in Singapore
MIRAILAB BIOSCIENCE Inc. (Headquarters: Chuo-ku, Tokyo; CEO: Megumi Tanaka) has partnered with Global Ocean Distribution Pte Ltd (Headquarters: Circular Road; Director: Getty Poon) to introduce its premium supplement brand MIRAI LAB, which features β-Nicotinamide Mononucleotide (β-NMN), to the Singapore market.…
MedLumics Completes FIM Study, to Present AblaView® PFA Results at AF Symposium 2025
Peter Hinchliffe, CEO of MedLumics, stated, “This marks a key milestone in validating a definitive Atrial Fibrillation (AF) ablation system, utilizing Pulsed Field Ablation (PFA) and Optical Coherence Reflectometry (OCR) to monitor and confirm ablation success in real-time under direct…

