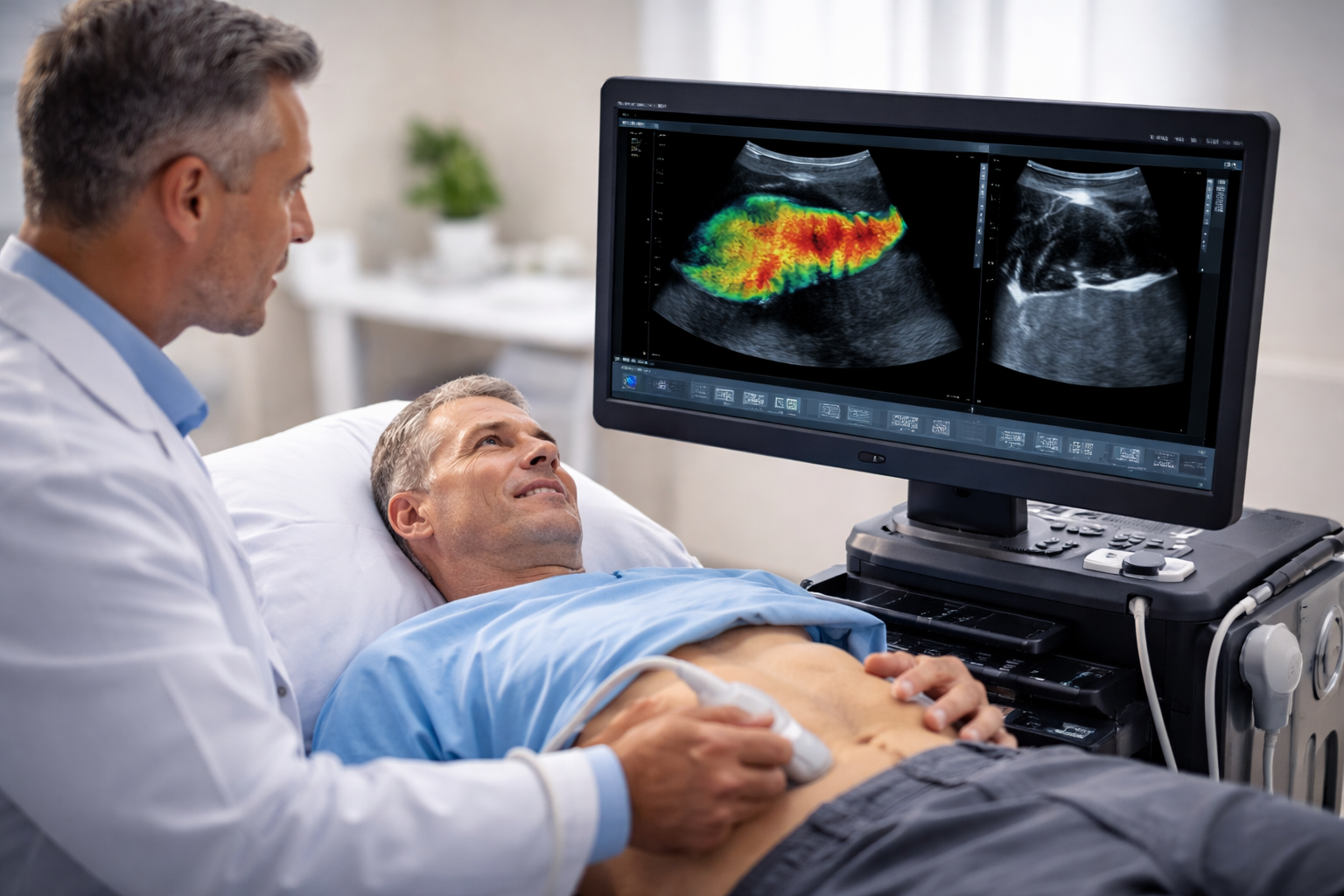
TAEUS® Demonstrates High Reliability in Clinical Study, Addressing Critical Diagnostic Needs
Why is the early detection and monitoring of steatotic liver disease (SLD) so challenging? ENDRA Life Sciences Inc. a pioneer in thermoacoustic biomarker imaging, has announced favorable results from a clinical study evaluating the measurement consistency of its TAEUS® Liver device. The study, which involved 14 subjects and 56 unique measurements, demonstrated the device’s robust intra-user repeatability and inter-user reproducibility, essential for long-term monitoring of Metabolic Dysfunction-Associated Steatotic Liver Disease (MASLD), a condition affecting approximately 30% of adults worldwide.
ENDRA’s TAEUS® Liver device achieved an Intraclass Correlation Coefficient (ICC) of 0.89 and a Standard Error of Measurement (SEM) of 3.3%, placing it in the “Good to Excellent” performance range. These results are significant because they show that TAEUS can deliver consistent and reliable results, similar to MRI-PDFF, but at a fraction of the cost and with greater accessibility. As Alexander Tokman, Chief Executive Officer of ENDRA Life Sciences, stated, “These new performance data strongly support our belief that TAEUS can deliver consistent results similar to an MRI, but at the point of patient care and at a much lower cost. This addresses a critical need for clinicians and pharmaceutical companies.”
Key Insights at a Glance
- Measurement Consistency: TAEUS® achieved an ICC of 0.89 and an SEM of 3.3%, indicating high reliability.
- Cost-Effective Solution: TAEUS® offers a more affordable alternative to MRI-PDFF for liver fat quantification.
- Clinical Relevance: The device’s performance supports its use in routine clinical practice and clinical trials.
- Market Impact: TAEUS® addresses the diagnostic needs of over two billion people affected by MASLD.
Why Reliable Liver Fat Measurement Matters
The ability to accurately and consistently measure liver fat is crucial for the early detection and monitoring of MASLD. Traditional methods like MRI-PDFF, while highly accurate, are expensive and not widely available, creating significant barriers for the two billion people affected by MASLD. The high cost and limited availability of MRI-PDFF make it impractical for frequent monitoring, which is essential for effective treatment and management of the disease. This gap in diagnostic tools has left clinicians and pharmaceutical companies in need of a more accessible and cost-effective solution.
The TAEUS® Solution: A Game-Changer in Liver Disease Monitoring
Just as a lighthouse guides ships through treacherous waters, TAEUS® guides clinicians through the complexities of liver disease diagnosis. The device’s high ICC and low SEM demonstrate its reliability and consistency, making it a viable alternative to MRI-PDFF. Unlike traditional ultrasound techniques, which suffer from high inter-observer variability and subjective interpretations, TAEUS® provides standardized, accurate, and repeatable liver fat measurements. This precision is crucial for both routine clinical practice and patient recruitment in clinical trials, where the ability to detect small, incremental changes in liver fat is essential.
ENDRA’s Commitment to Advancing Liver Disease Diagnostics
ENDRA Life Sciences is committed to addressing the critical need for reliable and accessible diagnostic tools in the fight against MASLD. The TAEUS® Liver device’s performance in the clinical study underscores its potential to revolutionize liver disease monitoring. By combining the precision of MRI-PDFF with the practical deployment advantages of ultrasound-based systems, TAEUS® offers a solution that can be deployed at the point of patient care. As new metabolic therapies enter the market, the ability to monitor treatment progress without the cost burden of repeated MRI scans becomes increasingly important.
Future Outlook
The future of liver disease diagnostics is evolving, and TAEUS® is at the forefront of this transformation. With its high reliability and cost-effectiveness, the device is poised to play a pivotal role in the management of MASLD. As ENDRA continues to refine and expand the capabilities of TAEUS®, the potential for widespread adoption in clinical settings and pharmaceutical research grows. The company’s innovative approach to thermoacoustic biomarker imaging is set to address a critical gap in the market, improving patient outcomes and supporting the development of new treatments.
Conclusion
ENDRA Life Sciences’ TAEUS® Liver device represents a significant advancement in the early detection and monitoring of MASLD. By providing a reliable, cost-effective, and accessible solution, TAEUS® addresses a critical need in the healthcare industry. For healthcare providers and pharmaceutical companies, this breakthrough offers a new tool to enhance patient care and clinical research. How will your organization leverage this technology to improve patient outcomes and advance medical research? Join the conversation in the comments below.
About ENDRA Life Sciences Inc.
ENDRA Life Sciences is the pioneer of Thermo Acoustic Enhanced UltraSound (TAEUS®), a ground-breaking technology being developed to assess tissue fat content and monitor tissue ablation during minimally invasive procedures, at the point of patient care. TAEUS® is focused on the measurement of fat in the liver as a means to assess and monitor steatotic liver disease and metabolic dysfunction-associated steatohepatitis, chronic liver conditions that affect over two billion people globally, and for which there are no practical diagnostic tools.
Source link: https://www.businesswire.com/